 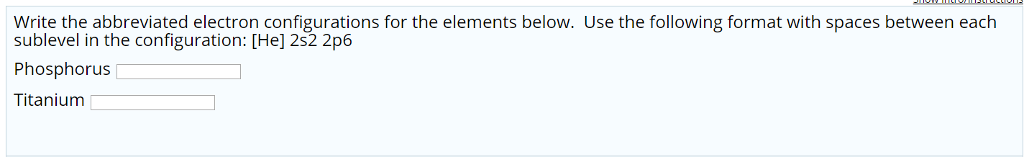
So in the third shell there's s, p ,and d orbitals which, if fully filled, do add up to 18 electrons. Sorry for the long response, but that's the stuff you need to know to do electron configurations well. The order which you fill subshells for more massive elements follows the aufbau principal. The first 1 is the shell number, the s is the subshell the electron exists in, the 1 superscript is the number of electrons in that subshell. Shell numbers are written first, then the subshell as a letter, then the number of electrons in that subshell are written as a superscript to the letter.įor for example neutral hydrogen has the electron configuration of: 1s^(1). Putting all these together you can specify exactly which electron you are referring to in an atom.įor the purposes of electron configuration though, we only care about the principal quantum number (the shell number), the angular quantum number (the subshell or the letter), and the number of electrons in those subshells which are basically a result of the magnetic quantum and spin quantum numbers. This is why orbitals can only hold a maximum of 2 electrons because they can only take on one of two ms values. This tells us why the s subshells only have 1 orbital while p subshells have 3, and the other subshells have the amount of orbitals that they do (and by extension how many electrons they can hold).įinally there is spin quantum number, represented by ms, of which an electron in an orbital can only take on two values: +1/2 or -1/2, broadly referring to the electron spinning clockwise or counterclockwise if you want to think of it like that. The values of ml in a subshell range from +l to -l. The magnetic quantum number, represented by ml, tells you the specific orbital within the subshell an electron is in. The values of l correspond to the subshell letters, so: l=0 is s, l=1 is p, l=2 is d, and l=3 is f. The subshell tells us broadly the shape the orbitals which hold the electrons take on and these are represented by the letters s, p, d, and f. Electrons in the second electron shell can have a value of l=0 AND 1 since n=2, and so on. So electrons in the first electron shell can only have a value of l=0 since n=1. The allowed values of l in an electron shell are determined by: l = n-1. The angular quantum number tells you the subshell an electron is in within an electron shell and is represented by the letter 'l' often. So elements in the first row of the periodic table have the first electron shell, or n=1, as their valence shell or outermost electron shell. The principal quantum number, often represented by n, is another term for the electron shell and are positive integer values (n=1,2,3,etc.) These electron shell numbers broadly correspond to the period, or row, an element is in.  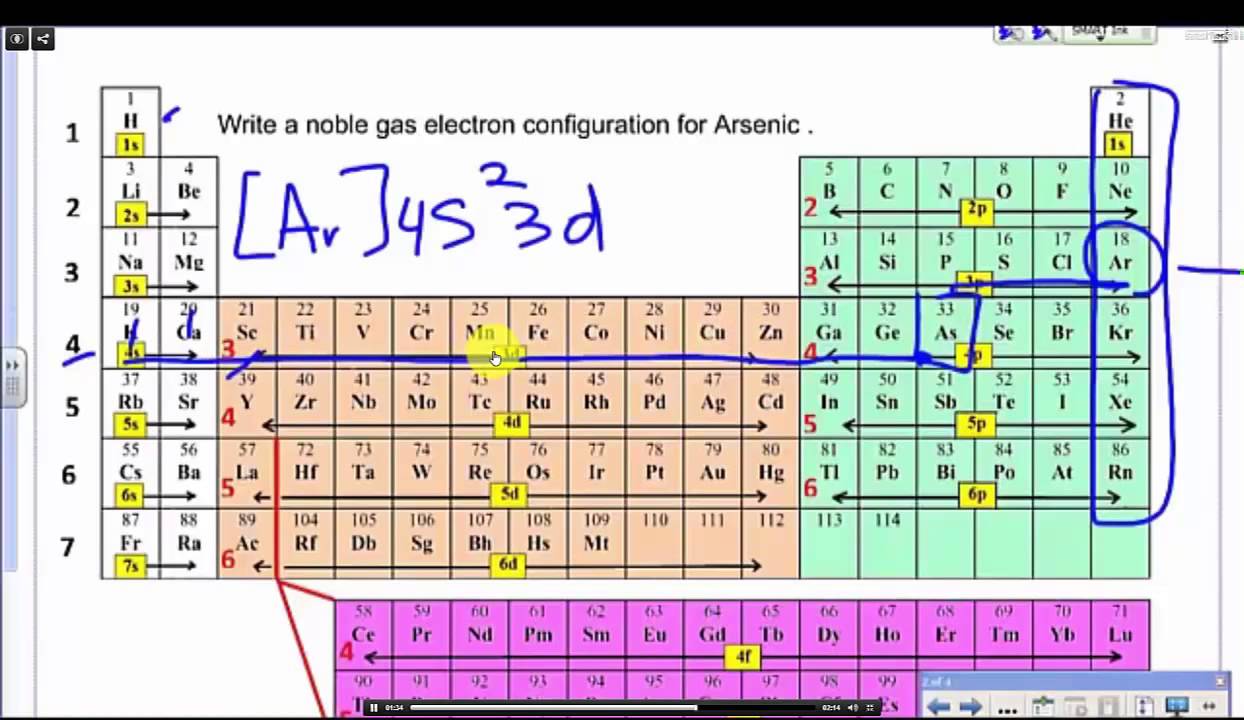
Due to the Pauli exclusion principal no two electrons can have exactly the same quantum numbers. These include the principal quantum number, the angular quantum number, the magnetic quantum number, and spin quantum number. These regions where we find electrons are represented by the quantum numbers, of which there are four. Such an arrangement helps explain the periodicity and periodic trends observed across the elements of the periodic table.So electrons exists in clouds in a way around the nuclei of atoms. The N shell containing 4s, 4d, 4p and 4f, can carry 32 electrons. The M shell contains 3s, 3p, and 3d, and can carry 18 electrons. The K shell contains a 1s subshell hence it can carry 2 electrons, the L shell has 2s and 2p, and can carry 8 electrons. This decides the electron capacity of the shells. The maximum electrons that can be carried by the sub-shell S is 2, by P is 6, by D is 10, and the F sub-shell can carry 14. Each shell and subshell have a limitation on the amount of electrons that it can carry. The subshells have a distinct shape and configuration, in which the electrons move freely. They stand for sharp (S), principal (P), diffuse (D), and fundamental (F). The shells are labeled K, L, M, N, and so on, from the innermost to the outermost shell.Įach shell has subshells that are named for the type of emission lines produced from different states of angular momentum. This model has been widely accepted, and according to it, each atom has shells, which further have subshells. It involves the specific arrangement of electrons in shells and sub-shells of Bohr’s atomic model. The concept of electronic configuration has replaced the older concept of valency and valence electrons. The electronic configuration of each element is decided by the Aufbau principle which states that the electrons fill orbitals in order of increasing energy levels. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
